Study design
A prospective case series study was conducted between 01/01/2018 and 31/06/2020. All patients provided informed consent to participate in the study, which was conducted in accordance with institutional standards.
Patient population
The patients were enrolled consecutively. The inclusion criteria were all adult distance runners diagnosed with iliotibial band syndrome and with a negative response to non-operative treatment after six months. Distance runners were defined as professional or amateur subjects completing medium (1500 m) and long (marathon and ultra-trail runners) distances.
The exclusion criteria were: (i) incomplete clinical reports; (ii) non-distance runners; (iii) additional injuries that interfere with running; (iv) bilateral involvement, (v) negative local anesthetic infiltration test; and (vi) revision surgeries after previous ITB procedures.
The patient must meet all inclusion criteria and none of the exclusion criteria. Before inclusion in the study, all patients completed a preoperative protocol regardless of additional tests performed up to that point.
Preoperative protocol
Complete medical history and physical examination were recorded in all patients. A local anesthetic infiltration test was performed, which consisted of an ultrasound-guided sub-iliotibial bursa infiltration with 2 ml of 2% mepivacaine, immediately followed by a 5 km race. If the patient’s symptoms were temporarily relieved during the race, the test was considered positive.
High-field MRI (≥ 1.5 T) was performed in all cases after the patient had exercised in the 72 hours before the scan, increasing the sensitivity of the imaging technique when edema appeared at the level of the LFC or ITB ( Figure 1 ).
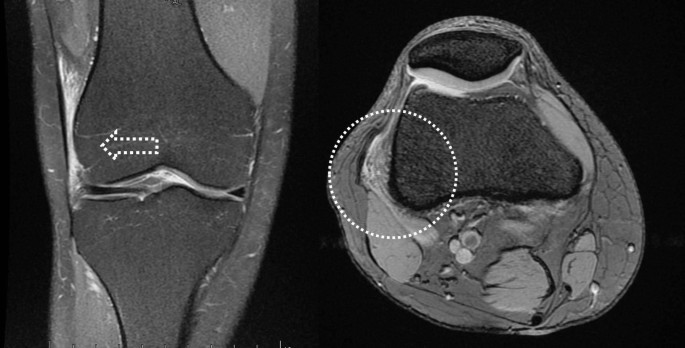
Preoperative MRI: coronal (right) and axial (left) images showing edema at the ITB.
Before the surgical indication, a specific rehabilitation program was performed to optimize conservative treatment with techniques not previously used in the patient, including fascia lata stretching exercises, proximal eccentric muscle training, intra-tissue percutaneous electrolysis and at least three focal shock wave exercises. sessions.
Independent variables and outcome variables
Demographic data (age, gender and body mass index -BMI-), comorbidities, athletic discipline, time to surgery and postoperative follow-up time were collected in all patients.
The intraoperative characteristics (time of ischemia, confirmation of ITBS, identification of concomitant lesions and need for drainage) and intraoperative and postoperative complications were also recorded.
The main variables of the study were the rate and time of return to the previous sports level, which were reported by patients during follow-up visits. Return to the previous sport level was considered a dichotomous outcome and was defined as participation after undergoing the PLAR technique in at least one race of the same distance as before the injury, at or above the pre-injury competitive level. The return to sport percentage was calculated from the number of athletes who returned to sport, from the number of athletes who underwent the PLAR technique, and expressed as a percentage.
The secondary variables were the clinical evaluation of the patients based on the Activity Rating Scale (ARS), the International Knee Documentation Committee (IKDC) questionnaire and the level of satisfaction. The results of the ARS and IKDC scales were interpreted as follows: excellent = 95–100 for IKDC and 15–16 for ARS; good = 84–94 for IKDC and 13–14 for ARS; and fair = 65–83 for IKDC and 10–12 for ARS. The level of satisfaction was evaluated in all patients with a poll based on the question: did the operation meet your expectations? The possible answers were: completely satisfied, largely satisfied, somewhat satisfied, dissatisfied.
Surgical procedure
All procedures were performed by the same surgeon. The ITBS diagnosis was confirmed intraoperatively by observing a collapse of the space between the LFC and the ITB due to a combination of bursitis and hard fibrotic adhesions that prevented the passage of the arthroscopy optic (Fig. 2).
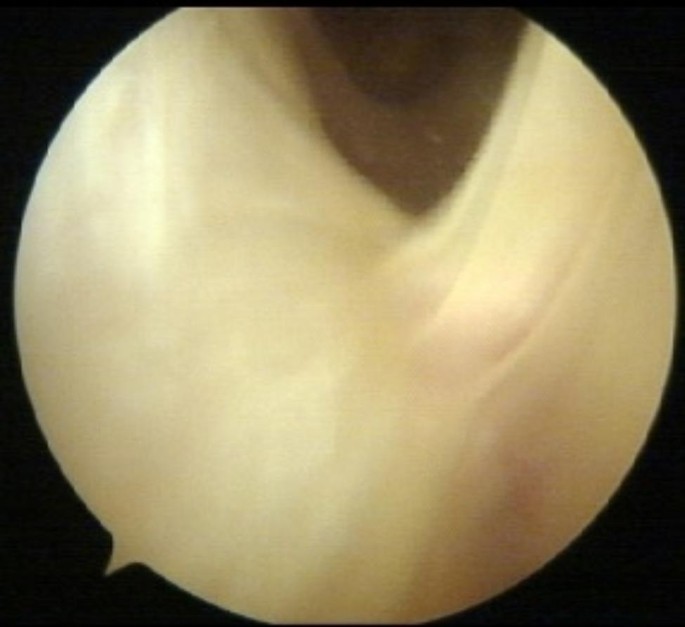
Intraoperative view. Fibrotic adhesions between the LFC and the ITB.
Patients were placed supine on a conventional table with arthroscopic support, during which an ischemia cuff was placed around the thigh and standard aseptic preparation was performed. The LFC, fibular head, Gerdy’s tubercle, and anteromedial (AM) and anterolateral (AL) standard portals were identified and marked.
The procedure began with routine diagnostic arthroscopy through the AL portal. If there was any doubt about additional lesions, an additional AM portal was used to allow tactile examination of the knee structures. Under direct intra-articular view, the superolateral (SL) portal was prepared using a 16G Abbocath spinal needle (Hospira, Lake Forest, IL, USA) as a guide, always passing through the tendon portion of the vastus lateralis muscle or the capsule, taking care not to perforate the quadriceps muscle tissue (Fig. 3). All portals were prepared with a No. 11 scalpel blade.
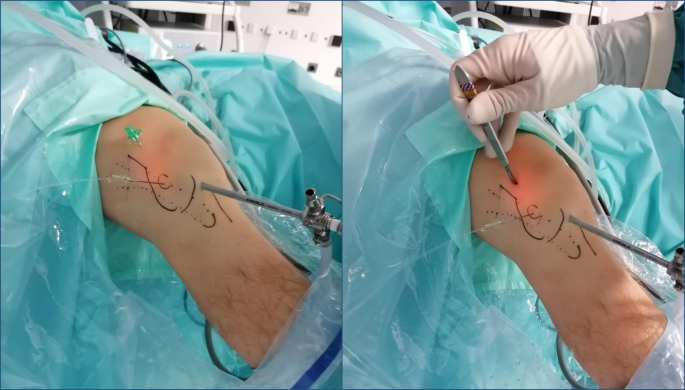
Intraoperative view. Superolateral portal (SLP) using a 16G Abbocath spinal needle as a guide
With the knee in 30° flexion, we initially performed debridement and resection of the lateral synovial recess, using a motorized shaver (Fig. 4) and a vaporizer (90 degrees, model 405Q3, Bonss Medical Tech, Taizhou, Jiangsu, China) (Fig. 5). In patients with ITBS, we can observe abnormal anatomy with increased fibrosis in the lateral synovial recess. Therefore, we consider it of utmost importance to perform a wide resection in this area until we obtain a complete view of the iliotibial band externally and the LFC medially, even including the external meniscal wall in the anterior half, and able are to pass the optic from the anterior to the popliteal tendon in the posterior zone, always preserving the meniscal-tibial and meniscal-femoral ligaments. This procedure was performed primarily from the SL portal under visual control from the AL portal, with reversal of the two portals to complete the release.
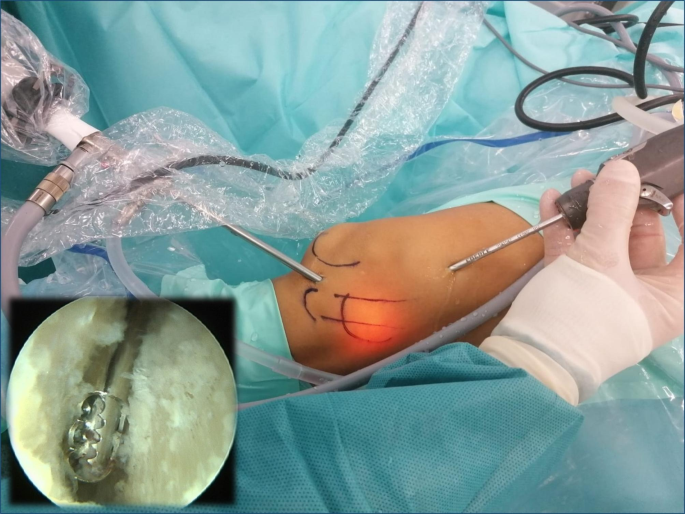
Intraoperative view. Loosening the fibrous adhesions in the space between the LFC and ITB using a motorized shaver
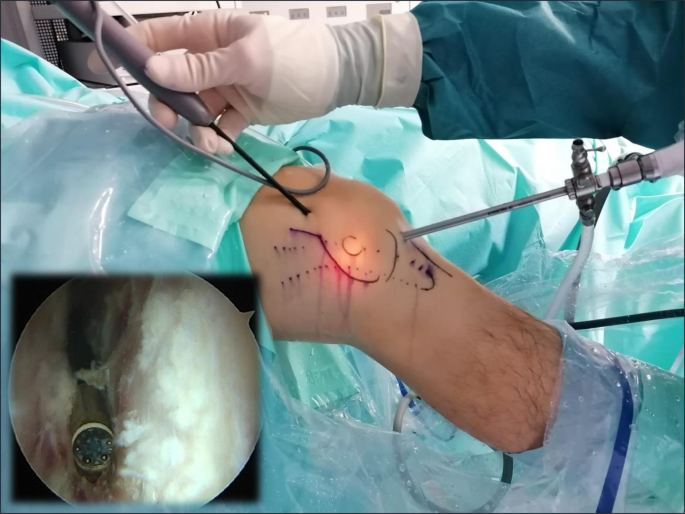
Intraoperative view. Releasing the fibrous adhesions in the space between the LFC and ITB using a vaporizer
The second part of the procedure involved the percutaneous lengthening of the ITB under direct vision by arthroscopy. This was done with controlled knee varus at 30° flexion, seeking a balance between extension and maintenance of muscle function. An 18G 3-mm needle scalpel (Nokor needle; Becton Dickinson and Co., Franklin Lakes, NJ, USA) was used to perform controlled micro-tenotomies as a micro-pie crust technique on the ITB. In all cases they were made longitudinally and parallel to the fibers, and in those cases with greater fibrosis of the ITB, the tenotomies were also made transversely in the posterior third (Fig. 6).
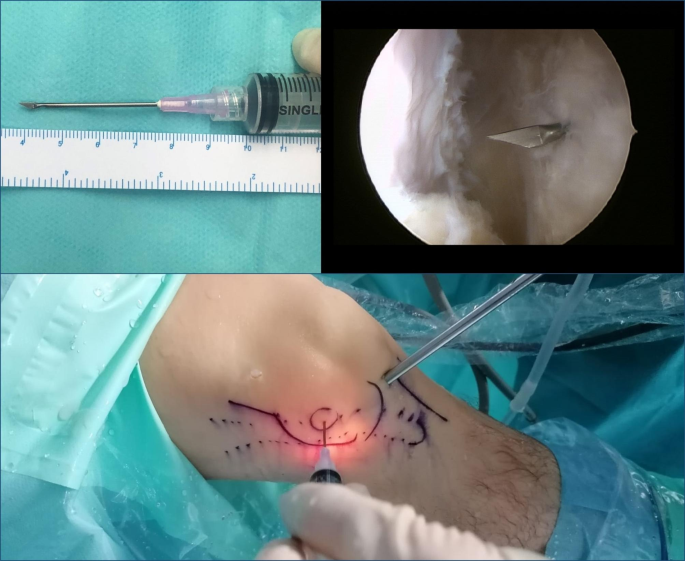
Intraoperative view. Micro-tenotomies on the ITB with an 18G 3 mm needle scalpel
After completion of the procedure, the skin was closed with Prolene (Ethicon, Inc.) 2/0, and a compressive elastic bandage was placed, with semi-rigid support in the external zone, where a bulge typically forms due to fluid extravasation via the microfibers. -tenotomies. Redon drainage (Fresenius Kabi AG, Bad Homburg, Germany) was used for 12 hours in patients with intraoperative identification of a sub-iliotibial bursa associated with significant vascular infiltration, and in all cases we infiltrated a mixture of corticosteroids and local anesthetic (2 ml Celestone Cronodose + 4 ml 2% mepivacaine).
Postoperative protocol
All patients were discharged with full weight bearing assisted by two crutches depending on tolerance.
Rehabilitation started from the first postoperative day. During the first two weeks, full joint range recovery exercises, isometric exercises, and even post-assisted squats were allowed to minimize muscle atrophy. Between weeks 2 and 4, eccentric muscle training (free, weight-bearing and single-foot squats, as well as frontal and lateral lunge exercises) combined with proprioception exercises using a BOSU ball (both sides up) or an unstable platform was allowed. From weeks 4 to 8, plyometric exercises, elliptical taping, and static cycling exercises were increased, and gentle jumping exercises were allowed depending on tolerance. From the 8th week onwards, and depending on the patient’s muscular and proprioceptive status, we allowed running a distance of 1 km every other day, combining walking and running exercises, and added distance or running exercises every two days. speed increases of 10% if tolerance was found. Good. From the 12th week after the operation, recovery was allowed to continue at the athletics club under the supervision of the coach or physiotherapist.
Follow-up protocol
A minimum follow-up of 12 months was performed. Postoperative data were collected in all patients at 15 days, 1, 3, 6 and 12 months and at the end of follow-up (medical discharge). Complications and clinical course were assessed at all visits, while sports performance and the ARS and IKDC questionnaires were assessed at 3, 6 and 12 months, without access to a copy of the scale during the intervening period, to avoid the patient himself – monitoring the recovery and influencing the final result. The level of satisfaction was recorded at the last follow-up visit.
static analysis
The statistical analysis was performed using the SPSS® version 22.0 package for Mac (IBM, NY, USA). Statistical significance was considered for p ≤ 0.05 and a statistical power of 90%.
Standard descriptive statistics including measures of central tendency (mean/median) and variance (standard deviation). [SD]/interquartile range [IQR]) were calculated, as well as frequencies and ratios.
The preoperative and final follow-up functional scores were compared using the Wilcoxon Signed-Rank test.
A multiple nonparametric analysis comparing the IKDCS and ACS scales preoperatively and at 6 and 12 months was performed using Friedman’s statistical test.