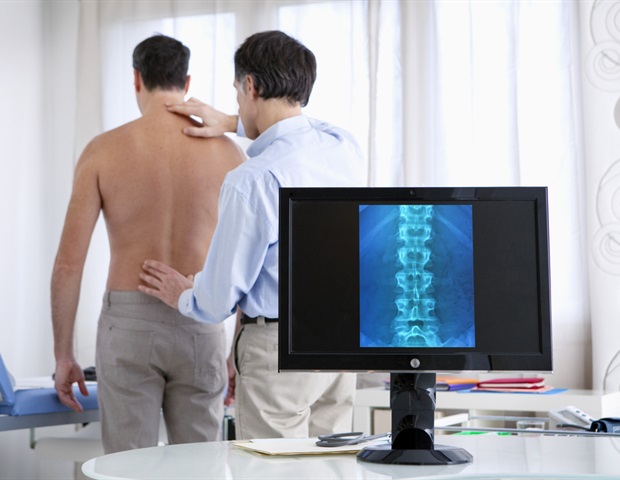
LEWISVILLE, Texas, November 6, 2023–(BUSINESS WIRE)–Orthofix Medical Inc. (NASDAQ:OFIX), a leading global spine and orthopedics company, today announced the full commercial launch in the U.S. of the WaveForm® L Lateral lumbar interbody system. Designed for lateral lumbar interbody fusion (LLIF) procedures, the 3D printed WaveForm L features a porous structure that prioritizes strength and stability to provide a robust fusion environment.
“Designed to safely and reproducibly treat the spine via indirect decompression and sagittal alignment restoration, the WaveForm L features a large core opening for the placement of bone graft material to optimize bone fusion throughout the body,” said Dr. Donald Blaskiewicz, director of Spinal Malformation at St. Luke’s Boise Medical Center in Boise, Idaho, and faculty at UCSD in San Diego, CA. “The WaveForm L also has enhanced imaging properties to assist with radiographic visualization during intra- and postoperative imaging. These combined features make it one of the best 3D printed interbodies available.”
WaveForm interbodies are uniquely created with a primary focus on structure, surface and stability. Constructed from a repeating wave-like structure, WaveForm L is designed to efficiently distribute compressive loads and provide high porosity for optimized stiffness without compromising strength.1,2 With an 80 percent body porosity, WaveForm L provides improved imaging properties and greater graft packability, while the optimized 65 percent endplate porosity of the wave-like structure provides bone ingrowth and early mechanical stability.3,4,5
“We are committed to delivering a comprehensive portfolio of innovative, procedurally focused products that are strategically designed to work together to drive fusion,” said Kevin Kenny, president of Orthofix Global Spine. “WaveForm L represents the latest developments in patented spinal implant technology, which is designed to address the many nuances of spinal pathology to meet the individual needs of patients, providing both clinical and economic value to patients, surgeons and hospital systems.”
Lateral lumbar interbody fusion (LLIF) procedures represent approximately 20 percent of the interbody device market. The number of these procedures is expected to continue to increase based on the rapid growth of titanium-coated PEEK devices and 3D printed metal devices. According to 2022 data, the LLIF market in the US is estimated at over $350 million.6
- Data available, TM-0043-22
- Kelly, Cambre N., et al. “Design and structure-function characterization of 3D printed synthetic porous biomaterials for tissue engineering.” Advanced Healthcare Materials 7.7 (2018): 1701095.
- Data available, TM-0071-23
- Data available, D0006845
- Kelly, C.N., Wang, T., Crowley, J., Wills, D., Pelletier, M.H., Westrick, E.R., Adams, S.B., Gall, K., & Walsh, W.R. (2021). High-strength, porous, additively manufactured implants with optimized mechanical osseointegration. Biomaterials, 279, 121206. https://doi.org/10.1016/j.biomaterials.2021.121206
- Data on file. Market estimates based on iData 2022 US Market Report Suite for Spinal Impants and MIS.
About Orthofix
Orthofix and SeaSpine merged in January 2023 to form a leading global spine and orthopedics company with an extensive portfolio of biologics, innovative spine hardware, bone growth therapies, specialty orthopedic solutions and a leading surgical navigation system. The products are distributed in approximately 68 countries around the world.
The company is headquartered in Lewisville, Texas, where it conducts general business, product development, medical education and manufacturing, and corporate offices in Carlsbad, CA, with a focus on spine and biologics product innovation and surgeon education, and Verona, Italy. with an emphasis on product innovation, production and medical education for orthopedics. The combined company’s global R&D, commercial and manufacturing footprint also includes facilities and offices in Irvine, CA, Toronto, Canada, Sunnyvale, CA, Wayne, PA, Olive Branch, MS, Maidenhead, UK, Munich, Germany, Paris, France and São Paulo, Brazil. For more information, visit Orthofix.com.
Forward-Looking Statements
This press release may contain forward-looking statements within the meaning of Section 21E of the Securities Exchange Act of 1934, as amended, and Section 27A of the Securities Act of 1933, as amended. In some cases, you can identify forward-looking statements by terminology such as “may,” “will,” “should,” “expects,” “plans,” “anticipates,” “believes,” “estimates,” “projects” , ‘intends’, ‘predicts’, ‘potential’, ‘continue’ or other similar terminology. Orthofix cautions you that statements in this press release that are not descriptions of historical facts are forward-looking statements based on the company’s current expectations and assumptions. Any forward-looking statement contained in this press release is subject to risks and uncertainties that could cause actual results to differ materially from those expressed or implied by such statement. Applicable risks and uncertainties include, but are not limited to: the ability of newly launched products to perform as designed and intended and to meet the needs of surgeons and patients, including due to the lack of robust clinical validation; and the risks identified under the heading “Risk Factors” in Orthofix Medical Inc.’s annual report. on Form 10-K for the fiscal year ended December 31, 2022, which was filed with the Securities and Exchange Commission (SEC) on March 6. , 2023. The Company’s public filings with the Securities and Exchange Commission are available at www.sec.gov. You are cautioned not to place undue reliance on forward-looking statements, which speak only as of the date on which they are made. Orthofix does not intend to revise or update any forward-looking statement contained in this press release to reflect events or circumstances occurring after the date of this press release, except as may be required by law.
Contacts
Media relations
Denise Landry
DeniseLandry@orthofix.com
214.937.2529
Investor Relations
Louisa Smith, Gilmartin Group
IR@orthofix.com