Study design and database
This was a descriptive, retrospective claims database study using the DATASUS database. The study identified patients with RA who sought care within SUS between January 1, 2010 and December 31, 2020.
The administrative claims data in DATASUS is presented as procedure codes from billing data and includes demographic information, all procedures (inpatient and outpatient), costs, and additional information23. Hospital admission (SIH [Sistema de Informações Hospitalares])24 and outpatient (SIA [Sistema de Informações Ambulatoriais])11 data exists separately and is linked at the patient level through multiple steps with different combinations of individual level information (date of birth, gender, city and zip code) for a probabilistic linkage approach. This Brazilian Healthcare Record Linkage (BRHC-RLK) methodology has been used in previous studies to enable a more comprehensive recording of each patient’s health record and thus allow a more complete evaluation of their journey through the SUS system.25. The method is based on multiple steps with different combinations of patient information from both databases, making it possible to identify or link patient data in both systems, while maintaining the anonymized nature of the database.
Patient-level data within DATASUS is anonymized and encrypted before being made available to researchers. DATASUS is publicly available and does not require further approval from ethics committees, according to Brazilian Ethics Resolution No. 510/2016.
Study population
The study population included patients with at least one claim of RA (according to the International Classification of Disease, 10th edition). [ICD-10] codes: M05.0, M05.3, M05.8, M06.0, M06.8, or M08.0) and ≥ 2 claims for disease-modifying antirheumatic drugs (DMARDs) ≥ 1 month apart in the 2010 survey– 2020 period. This study examined a treated population where the index date was the first DMARD claim and followed until the end of the study period (December 31, 2020) or the last available information. Detailed DMARD definitions can be found in Supplementary Table S1.
To capture initial treatment and address the potential for misclassification common in claims data, patients with a DMARD claim without an RA ICD-10 code 12 months prior to the index date were excluded. The index date was defined as the date of the first RA ICD-10 and DMARD prescription in the public health system during the study period. Patients with RA with less than six months of follow-up were excluded, in an effort to reduce the number of individuals with a false diagnosis or lack of follow-up in the database.
Because SUS is a healthcare system with universal coverage, patients with additional private health insurance can also receive medications (such as expensive drugs) covered by SUS at no out-of-pocket cost. This is often observed in other therapeutic areas26. For this reason, we stratified the results across the following cohorts: Cohort 1 is the entire study population, Cohort 2 is SUS-exclusive (i.e., dependent on SUS for all healthcare-related encounters, procedures, and treatments), and Cohort 3 represents SUS + private patients ( i.e. depending only on SUS for prescription drug coverage)26.
Measurements of DMARD treatment
DMARD treatments measured using procedure codes (see Supplementary Table S1) were grouped into the following categories: csDMARD for conventional synthetics and/or immunosuppressants (ciclosporin, cyclophosphamide, chloroquine, hydroxychloroquine, leflunomide, methotrexate, azathioprine and sulfasalazine), bDMARD for biological drugs (adalimumab, abatacept, etanercept, infliximab, rituximab, tocilizumab, golimumab, certolizumab) and tsDMARD for a synthetic, oral target therapy [Janus kinase (JAK) inhibitor, tofacitinib].
Treatment patterns were evaluated by specific drug (independently of monotherapy or in combination) as provided for RA treatment and the order of available treatments in SUS, by line of therapy (LOT), time point of each drug, previous and subsequent DMARD treatments in SUS. The first treatment was the first therapy from the admission according to RA ICD-10 code. LOT was defined as at least three claims (dispensation) of the same drug (b/tsDMARD) in a row. A new series of at least three claims (dispensation) of the same drug in the correct order was considered a new line of treatment. Thus, the switch to a treatment was identified as at least three claims for drugs other than the previous one, which are not included in the definition of drugs used in combination. Gaps were allowed regardless of time and did not constitute a new LOT. First-line (LOT1) refers to initial treatment, first b/tsDMARD claim of RA during the study period. Second-line (LOT2) refers to the second b/tsDMARD used for RA treatment, when the first b/tsDMARD was stopped. Third line (LOT3) refers to the third b/tsDMARD, when the previous b/tsDMARD was terminated. csDMARDs used before b/tsDMARD were also assessed. Switching treatment was defined as at least three claims for drugs different from the previous one (new LOT), and not part of drugs used in combination.
static analysis
Derived variables included age and distance to the clinic. Age was defined as the age at the first claim of an ICD-10 code for RA in the database. Distance was calculated as the Euclidean distance (km) between two zip codes: the patient’s place of residence and the health care facility or tomography or antiangiogenic treatment facility, as applicable. Treatment switch, discontinuation and end of follow-up were the main outcomes of censoring events of interest, also relevant in defining LOT and creating Sankey diagrams.
Continuous variables (e.g., age) are summarized by central tendency (means, medians) and dispersion (variance, range); and for categorical variables (e.g. gender) based on absolute number and percentage. Stratifications and/or sensitivity analyzes were performed to evaluate differences in gender, age groups, patient region of residence, drug use, treatment line, and others.
Stratified analyzes for mainstream and new users were prespecified, and for SUS-exclusive and SUS+ private cohorts. Frequent users were patients with RA who were currently receiving bDMARD treatment, and new users were patients with RA who were starting a new bDMARD treatment (i.e., their first prescription). To describe the use and sequential patterns of RA bDMARD treatments, patients were stratified by treatment type, LOT-specific drug, and SUS-exclusive status.
In multivariable logistic regression analyses, age, SUS-exclusive status, distance to clinic (160+ km), and pre-index cs/imsDMARD and other independent predictors were included to evaluate initiated therapy (LOT1) with b/tsDMARD (JAKi). Multivariable analyzes were performed using Cox regression models evaluating predictors by time to switch to tsDMARD (JAKi) compared to bDMARD (LOT2+), applying the same independent predictors from multivariable logistic regression analyzes (age, SUS-exclusive status , distance to clinic, pre-index cs/imsDMARD, other), plus the number of previously used bDMARDs. Sankey diagrams were used for visualizations of treatment patterns. Sankey diagrams quantitatively illustrate the sequence of treatment (and/or duration of treatment) and allow stratification by subpopulations of interest with censoring based on different treatment, discontinuation, or end of follow-up. Kaplan Meier survival analyzes and plots were generated for time to switch from LOT1 to LOT2, among those treated with b/tsDMARD, analyzed by drug type and by SUS-exclusive status.
The visual representation of the time-to-event of the switch from LOT1 to LOT2 in patients receiving b/tsDMARD therapies was presented in Kaplan-Meier curves. The last available patient information or end of study period was considered censored for patients who did not switch from LOT1 to LOT2.
All analyzes were performed using Python version 3.6.9 and statistical significance was set at p < 0.05.



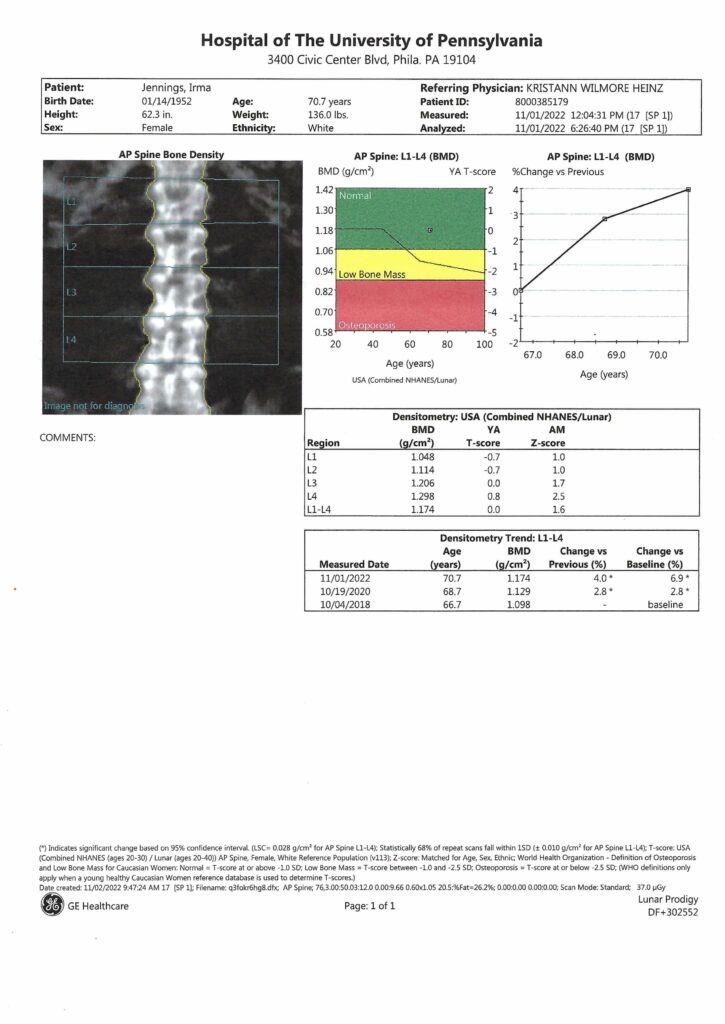
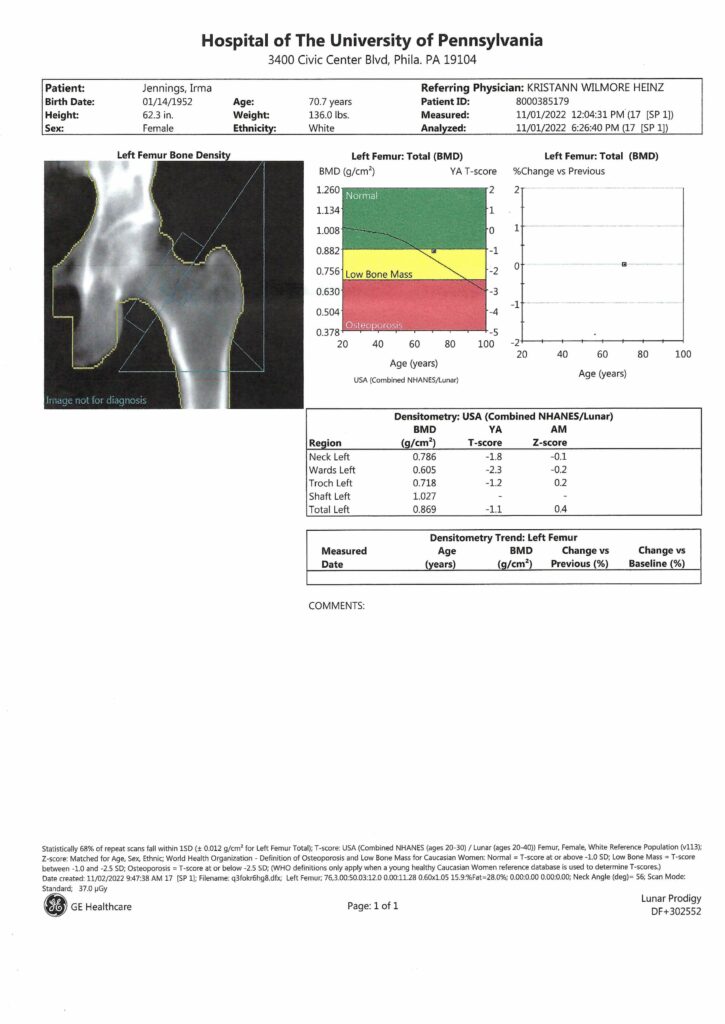
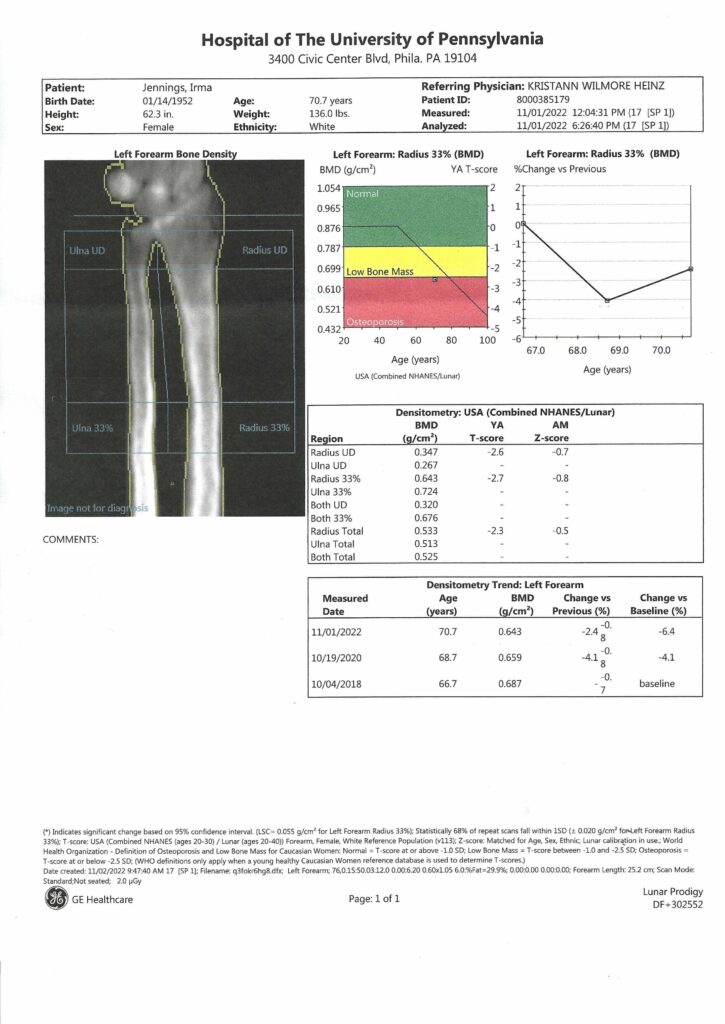
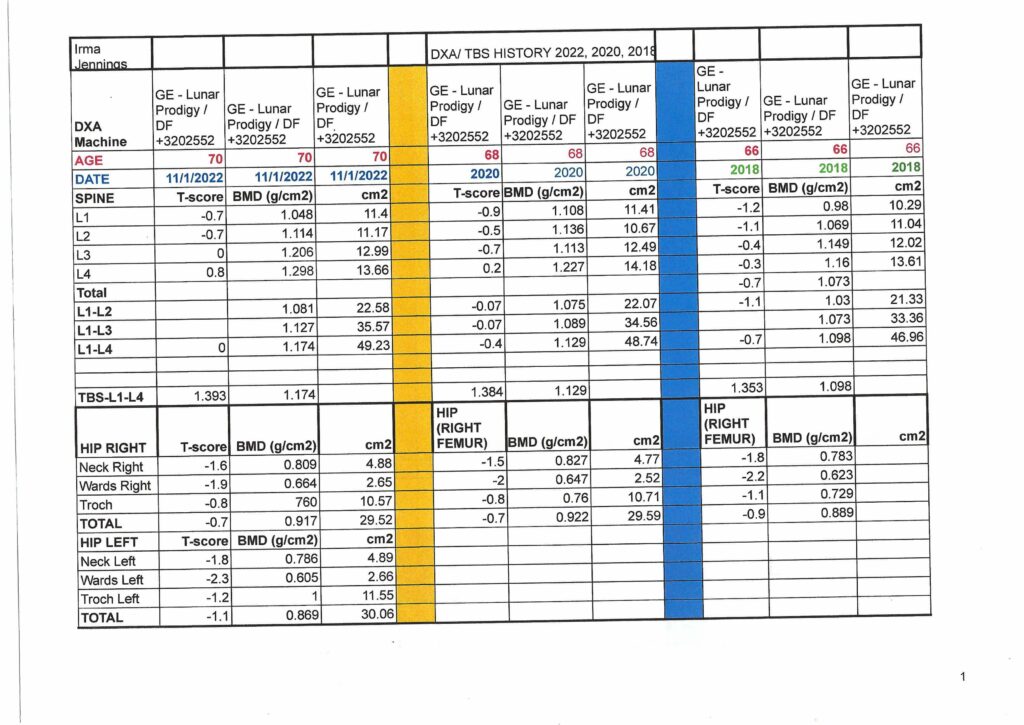




 This article was adapted from khn.org, a national newsroom that produces in-depth journalism on health issues and is one of the core operating programs at KFF – the independent source for health policy research, polling and journalism.
This article was adapted from khn.org, a national newsroom that produces in-depth journalism on health issues and is one of the core operating programs at KFF – the independent source for health policy research, polling and journalism.




