Recruiting a panel of leading KOL surgeons who believe Nociscan can improve the diagnosis and treatment of discogenic low back pain is a critical step in establishing Nociscan as the standard of care.
Aclarion’s KOL panel represents spine surgeons at some of the largest and most influential academic centers and private practices in the country, including leaders of national associations that advocate for protocols to improve clinical treatments.
The company plans to activate MRIs for every KOL surgeon to expand Nociscan access for their patients and demonstrate improvements in clinical outcomes to payers.
Aclarion will release initial scan volume data before the end of the year and begin reporting on quarterly scan volume in 2024, as increasing scan volume is a measure of increasing revenue and the likelihood of a coverage decision the local payer.
BROOMFIELD, CO, November 8, 2023 (GLOBE NEWSWIRE) — via NewMediaWire – Aclarion, Inc., (“Aclarion” or the “Company”) (Nasdaq: ACON, ACONW), a healthcare technology company that uses biomarkers and proprietary enhanced intelligence algorithms to help physicians identify the location of chronic low back pain, today announced that they have successfully achieved their goal of enrolling ten leading spine surgery KOLs to join the company help bring Nociscan to the standard of care for identifying discogenic low back pain.
“Since executing our IPO last year, Aclarion has consistently delivered value creation catalysts that fall within a very concise framework of technology readiness, clinical evidence and surgeon advocacy and are proven to bring disruptive technological advances to the standard of care in the healthcare industry. said Brent Ness, Chief Executive Officer of Aclarion. “The steps we have taken in a short period of time demonstrate that our actions follow our stated intentions. We are incredibly proud of the KOL panel that has worked to bring Nociscan to the standard of care.”
A cornerstone of the company’s strategy is Key Opinion Leaders (KOL) advocacy. Aclarion has now achieved its goal of engaging 10 leading spine physicians who are deploying Nociscan in their practices to close a major gap with conventional lumbar MR imaging, which cannot distinguish between pain and aging. Our KOL advisors are focused on advancing value-based healthcare through improved outcomes while advocating for societies and payers that leverage patient data and peer-reviewed, published clinical evidence. “KOL’s interest in Nociscan directly reflects the challenge of diagnosing and treating low back pain, and we are pleased that these physicians are advancing spine care with Nociscan,” said Ryan Bond, Chief Strategy Officer of Aclarion.
KOLs, consultants and advisors now include:
- Chris Ames, MD; University of CA San Francisco
- Gregory Basil, MD MBA; University of Miami
- Sigurd Berven, MD; University of CA San Francisco
- George Frey, MD; Advent Health Colorado
- Roger Hartl, MD; Weill Cornell Brain and Spine Center, New York, NY
- Dean Karahalios, MD; Advocate for the Aroura Health System
- James Keller, MD; University of Michigan Health West
- Alpesh Patel, MD MBA; Northwest
- Eric Potts, MD; St. Vincent’s, Ascension Day
- Juan Uribe, MD; Barrow Neurological Institute
- Bob Eastlack, MD; Scripps (surgeon consultant)
- Timothy Ryken, MD; (Consultant)
- Lawrence Tannenbaum, MD; RadNet (MD advisor)
- Jeffrey Lotz, PhD; University of CA San Francisco (Scientific Advisor)
The company will announce expansion of MRI access for each KOL once their MRIs are onboard. Dr. Frey and Hartl already have access to Nociscan and have used it in their clinical practice. As access to MRI increases, so will the potential scan volume, peer-reviewed evidence, and revenue.
For more information about the BEST trial, please visit: www.besttrial.org
For information about BACPAC, please visit: https://heal.nih.gov/research/clinical-research/back-pain
For more information about REACH, please visit: www.bacpac-reach.org
For more information about our published evidence, please visit: www.aclarion.com
For information about accessing Nociscan, please visit: www.aclarion.com
About Aclarion, Inc.
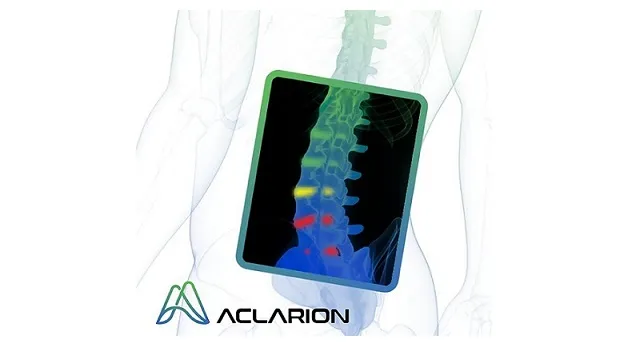
Aclarion is a healthcare technology company that uses magnetic resonance spectroscopy (“MRS”), proprietary signal processing techniques, biomarkers and enhanced intelligence algorithms to optimize clinical treatments. The company is entering the chronic low back pain market for the first time with Nociscan, the first evidence-based SaaS platform that helps physicians non-invasively distinguish between painful and non-painful discs in the lumbar spine. Through a cloud connection, Nociscan receives magnetic resonance spectroscopy (MRS) data from an MRI machine for each lumbar disc being evaluated. In the cloud, proprietary signal processing techniques extract and quantify chemical biomarkers shown to be associated with disc pain. Biomarker data is fed into proprietary algorithms to indicate whether a disc may be a source of pain. When combined with other diagnostic tools, Nociscan provides critical insights into the location of a patient’s low back pain, giving clinicians clarity to optimize treatment strategies. For more information please visit www.aclarion.com.
Forward-Looking Statements
This press release contains forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995, Section 27A of the Securities Act of 1933 and Section 21E of the Securities Exchange Act of 1934 about the Company’s current expectations about future results, performance, prospects and opportunities. Statements that are not historical facts, such as “anticipates,” “believes” and “expects” or similar expressions, are forward-looking statements. These forward-looking statements are based on management’s current plans and expectations and are subject to a number of uncertainties and risks that could materially affect the company’s current plans and expectations, as well as its future results of operations and financial condition. These and other risks and uncertainties are discussed in more detail in our filings with the Securities and Exchange Commission. Readers are encouraged to read the section entitled “Risk Factors” in the Company’s April 21, 2022 Prospectus as filed with the Securities and Exchange Commission on April 25, 2022 under Rule 424(b)(4), as well as other disclosures. included in the Prospectus and subsequent filings with the Securities and Exchange Commission. Forward-looking statements in this announcement are made as of this date and the Company undertakes no obligation to publicly update or revise any forward-looking statements, whether as a result of new information, future events or otherwise.
Investor contacts:
Kirin M. Smith
PCG Advice, Inc.
646.823.8656
ksmith@pcgadvisory.com
Media contacts:
Jodi Lamberti
SPRIG advice
612.812.7477
jodi@sprigconsulting.com