This is evident from a recent study published in the journal JCI Insight, the authors built on their previous work examining the anti-autoimmune disease effects of 6-gingerol, the most abundant phytochemical produced by the roots of the ginger herb. Because their previous work showed that this plant extract could reverse the effects of neutrophil hyperactivity in mouse model systems, the researchers herein evaluated whether oral consumption of whole ginger extracts could have similar effects both in mouse models and in human pilot trials. Their results show that ginger consumption for just seven days neutralizes hyperactivity in neutrophils in both in vivo systems. When taken by healthy individuals, it increases their resistance to developing conditions, including lupus and antiphospholipid syndrome (APS).
 Study: Ginger intake suppresses extracellular neutrophil formation in autoimmune mice and healthy humans. Image credits: Nataly Studio / Shutterstock
Study: Ginger intake suppresses extracellular neutrophil formation in autoimmune mice and healthy humans. Image credits: Nataly Studio / Shutterstock
Neutrophilic autoimmune diseases and the untapped potential of herbs
Antiphospholipid antibody syndrome (APS) is an autoimmune disease that mainly affects women between the ages of 30 and 40. APS results in the formation of abnormal proteins that promote clot formation in veins and arteries and is especially harmful to both mother and her fetus during pregnancy. APS and lupus, a frequent comorbidity characterized by circulating immune complexes that damage organs after their deposition, are lifelong, incurable conditions resulting from genetics, environmental exposure, or a combination of these. Both diseases result in significant mortality, morbidity and healthcare costs.
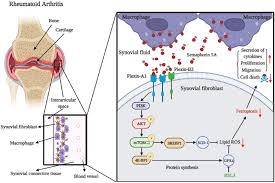
Previous research has shown that, despite having very different clinical profiles, both APS and lupus are pathologically caused by the exaggerated and abnormal formation of extracellular neutrophils, medically termed ‘NETosis’. During NETosis, neutrophils overexpress and secrete their nuclear chromatin in the form of web-like structures with pro-inflammatory properties and potentially harmful granule-derived proteins that, despite being localized in organs and the circulatory system, have serious consequences on the health.
Recent studies have shown that excessive NETosis, in addition to its own negative consequences, can result in sustained formation of autoantibodies, resulting in other autoimmune diseases that would otherwise have been suppressed by the body’s adaptive immune tolerance. Considering that most of these autoimmune diseases are incurable, require constant and usually expensive medical interventions, and carry significant mortality costs, finding a low-cost therapy for NETosis is imperative.
In recent years, scientific attention has shifted to the potential of herb-derived phytochemicals with anti-inflammatory properties as a wealth of safe and natural remedies against autoimmune diseases in general, and NETosis in particular. In a previous study, the authors of the current work showed that a purified ginger extract, 6-gingerol, shows promise in stimulating intracellular cyclic adenosine monophosphate (cAMP) and attenuating neutrophil phosphodiesterase (PDE) activity, both of which are important mechanistic results are from NETose.
In particular, their research found that NETs and neutrophils in manifestations of thromboinflammatory diseases not only influence APS, lupus and similar autoimmune diseases, but also promote adverse outcomes in communicable diseases such as coronavirus disease 2019 (COVID-19).
About the study
The current study aimed to determine whether whole ginger extracts have similar NETosis-reversing effects as 6-gingerol and have beneficial effects on consumption even for healthy individuals who do not exhibit autoimmune symptoms. This study represents a pilot study that may form the basis for future clinical testing of ginger’s beneficial proteins in the treatment of a spectrum of NETosis-related autoimmune diseases, including APS, lupus, vasculitis, rheumatoid arthritis, and even COVID-19.
Researchers began testing the efficacy of powdered whole ginger obtained from Aurea Biolabs (Kerala, India) in in vitro testing. Immunoglobulin G (IgG) was obtained from both APS and lupus patients (cases) and healthy controls and purified using the Protein G Agarose Kit (Pierce). The purity and concentration of IgG were estimated using Coomassie staining and BCA protein assay, respectively. For NETosis assays in human neutrophils, blood was collected from healthy human volunteers, purified via density gradient centrifugation, and neutrophils were isolated using dextran sedimentation and red blood cell (RBC) lysis. Flow cytometry and nuclear morphology microscopy were used to verify purity.
For NETosis assays, the above purified neutrophils were mixed with neutrophils derived from APS and lupus patients (three volunteers, respectively). In vitro assays consisting of immunofluorescence microscopy, measurements of phosphodiesterase (PDE) activity, and calculations of intracellular cAMP levels.
In vivo APS models included venous thrombosis experiments performed on male C57BL/6 mice (10-13 weeks). Female BALB/c mice (9 weeks old) were used for lupus testing. The venous thrombosis testing was performed using an electrolytic inferior vena cava (IVC) model. Circulating myeloperoxidase (MPO)-DNA complexes were then quantified and isolated thrombi were processed via thrombus section and immunohistochemistry.
Finally, the pilot study was conducted in humans. Participants older than 18 years and without NETosis-associated autoimmune diseases were recruited. Female participants had to be fertile to allow the evaluation of ginger consumption in combination with contraception. Women who were pregnant, lactating, or suffering from cardiovascular disease, diabetes, or cancer were excluded to avoid confusion due to their illness or the medications they were taking. All statistics performed in this study were 1-way analysis of variance (ANOVA) corrections based on multiple corrections.
Findings of the study
The main findings of this study were that ginger consumption significantly inhibited NETosis in healthy study participants, even after stimulation (mixing) of neutrophils from APS or lupus patients. This was expected given that their previous research reported the same findings on the use of purified 6-gingerol supplementation, and whole ginger extracts contained approximately 20% 6-gingerol. Similarly, this study presents that consumption of whole ginger inhibited cAMP-specific PDE activity, confirming previous findings.
In in vivo mouse models, consumption of whole ginger APS was shown to attenuate IgG-mediated venous thrombosis and NETosis. Remarkably, consumption of whole ginger attenuates lupus-relevant disease activity even in lupus-positive female BALB/c mice.
Pilot human experiments confirm that ginger is an important focus in future clinical trials, as it was found to stimulate neutrophil cAMP and reduce NETosis in healthy human volunteers, even after just seven days of ginger diet supplementation. To verify these results and confirm that the findings were not a byproduct of the small sample size (N = 9; 3 men, 6 women), the study was repeated with an unrelated volunteer cohort (N = 8). findings consistent across replications. Furthermore, the second cohort study revealed a reduction in plasma NET levels (measured by the MPO-DNA complexes).
Conclusion
The current study highlights the potential of whole ginger consumption as a safe and natural intervention, both to treat existing cases of APS, lupus and other NETosis-associated diseases and to prevent the development of these conditions in previously healthy people . They combined in vitro testing with in vivo mouse and human models and found that consumption of whole ginger attenuates venous thrombosis (APS) and significantly reduces the clinical features of lupus in mouse models.
In healthy human volunteers, ginger consumption for just seven days was associated with a notable reduction in NETosis and cAMP, confirming its potential against autoimmune diseases in future clinical trials.
“…we found that the dissolved ginger extract counteracted the PDE activity of neutrophils. The result was increased neutrophil intracellular cAMP levels, which were associated with blunted NETosis by human neutrophils in vitro. Such data complement recent studies that have reported a role for ginger extracts, and in particular 6-gingerol, as inhibitors of cAMP-specific PDE activity. Importantly, the suppressive effects of ginger on NETosis can be attenuated by blocking PKA activity, a key downstream cAMP-dependent kinase. The fact that increasing neutrophil cAMP and activating PKA would be beneficial for disease activity in mice is in good agreement with our previous work demonstrating the potential therapeutic target of this pathway in APS and lupus models with synthetic PDE4 inhibitors. ”
Magazine reference:
- Ramadan A. Ali, Valerie C. Minarchick, Miela Zahavi, Christine E. Rysenga, Kristin A. Sturm, Claire K. Hoy, Cyrus Sarosh, Jason S. Knight, M. Kristen Demoruelle. Ginger intake suppresses extracellular neutrophil formation in autoimmune mice and healthy humans. JCI Insight. 2023;8(18):e172011, DOI – https://doi.org/10.1172/jci.insight.172011, https://insight.jci.org/articles/view/172011