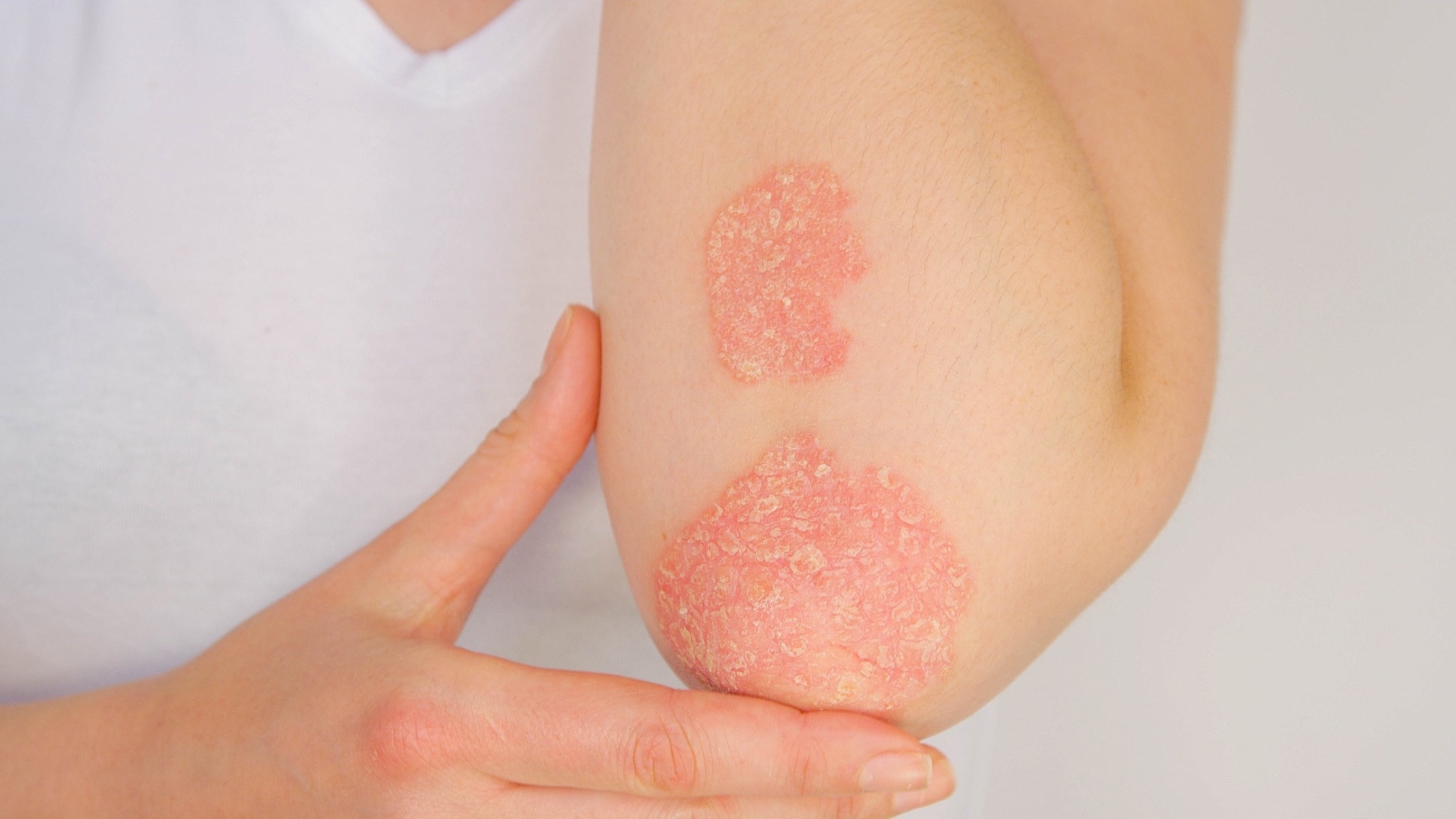
In a recent study published in the Scientific Reports Journal, researchers evaluated the risk of new autoimmune diseases in individuals with early psoriatic disease.
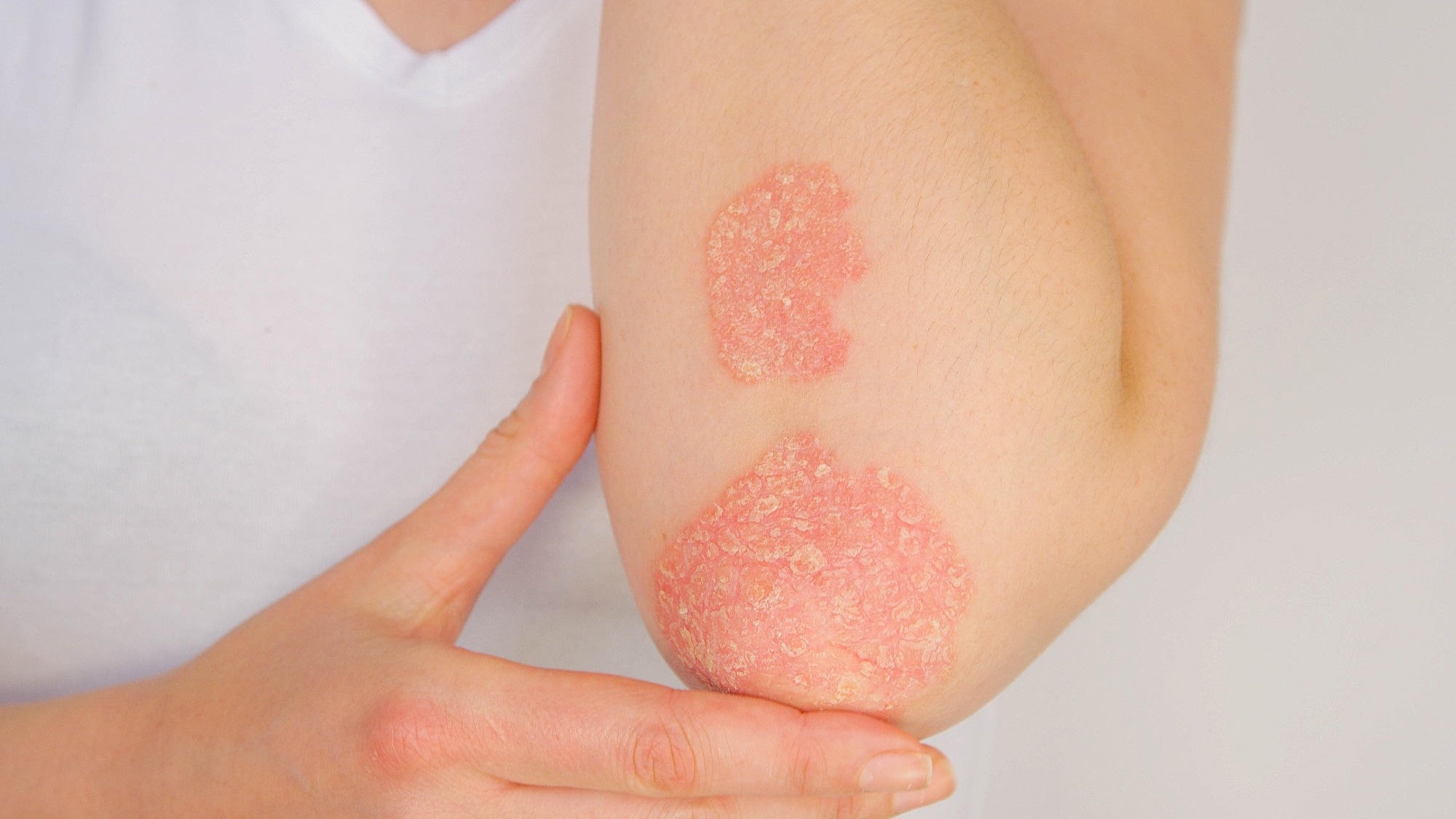 Study: Risk of incident autoimmune diseases in patients with newly diagnosed psoriatic disease: a national population-based study. Image credits: Flystock/Shutterstock.com
Study: Risk of incident autoimmune diseases in patients with newly diagnosed psoriatic disease: a national population-based study. Image credits: Flystock/Shutterstock.com
Background
Psoriatic disease, which often includes psoriasis and psoriatic arthritis, is a systemic inflammation-related disease with serious clinical consequences.
Autoreactive T lymphocytes, which express proinflammatory cytokines such as interleukins (ILs)-17 and 22, and interferon-gamma (INF-γ) characterize psoriatic diseases.
Studies have linked the autoimmune component of psoriasis to several autoimmune diseases, including autoimmune thyroid diseases, inflammatory bowel disease, alopecia areata, and autoimmune rheumatic diseases.
About the study
In the national population study, researchers evaluated the link between autoimmune diseases and psoriatic diseases.
Patients with newly diagnosed psoriatic disease between January 2007 and December 2019 were included in the study, using the Korean National Health Insurance Service (NHIS) database.
The team used previously established diagnostic algorithms for psoriatic disease in Korea to identify people with the condition. Patients with psoriatic disease had one or more recorded visits with psoriatic diseases such as psoriasis and psoriatic arthritis as their primary diagnostic code and vitamin D prescriptions.
Autoimmune diseases such as CD, UC, Graves’ disease, Hashimoto’s disease, SLE, RA, Sjögren’s syndrome, systemic sclerosis, AS, type 1 diabetes and alopecia areata were studied.
Comparators who had no diagnostic code for psoriatic diseases between January 2005 and December 2019 and no diagnostic code for autoimmune diseases during the washout were randomly selected and matched on sex and age at a 1:1 ratio.
In addition to the diagnostic codes, the researchers also added relevant prescription information for medications and RID codes for all diseases to the diagnostic algorithms for outcomes.
The team added follow-up information on newly identified autoimmune diseases from 2007 to 2020. Multivariate Cox regression modeling was performed to determine adjusted risk ratios (aHRs).
The Charlson Comorbidity Index (CCI) values for comorbidities such as hypertension, diabetes, dyslipidemia, chronic obstructive pulmonary disease (COPD), liver cirrhosis, chronic kidney disease, alcoholic liver disease, and heart failure were determined. The team conducted subgroup analyzes based on gender, age and severity of psoriatic disease.
Patients who received diagnostic codes for psoriatic disease between January 2005 and December 2006 were excluded from the study.
Additionally, the team excluded individuals diagnosed with autoimmune diseases [Crohn’s disease (CD), ulcerative colitis (UC), Graves’ disease, Hashimoto’s disease, systemic lupus erythematosus (SLE), rheumatoid arthritis (RA), Sjögren’s syndrome, ankylosing spondylitis (AS), systemic sclerosis, alopecia areata, and diabetes type 1] before diagnosis of psoriatic disease and those followed for less than one year.
Results
The study included 321,354 individuals in the psoriatic and control groups with a mean age of 43 years, and 59% were men.
Patients with psoriasis showed a significantly increased risk of ankylosing spondylitis, systemic lupus erythematosus, ulcerative colitis, Crohn’s disease, alopecia areata, type 1 diabetes and rheumatoid arthritis, with aHR values of 2.3, 1.9, 1.7 , 2.0, 1.4, 1.2 and 1.6. respectively.
On the other hand, the risks for Hashimoto’s disease, Graves’ disease, systemic sclerosis and Sjögren’s syndrome did not differ significantly between the groups. Type 1 diabetes, alopecia areata, AS, RA, SLE, UC, and CS had NNH values of 9,567, 1,295, 9,946, 3,256, 47,987, 17,899, and 39,988 individual years, respectively.
With the exception of type 1 diabetes, all autoimmune diseases showed a significantly increased risk in psoriatic subjects compared to controls after controlling for CCI and insurance type. After controlling for CCI and insurance type, the risk of type 1 diabetes (aHR, 1.1) was not significant in men with psoriatic disease compared with male controls.
After adjusting for type of insurance and CCI, all autoimmune diseases showed significantly greater risks in younger psoriatic individuals under 40 years of age and in individuals 40 years of age and older compared with controls.
After controlling for insurance type and CCI, CD risk (aHR, 1.4) was not significantly greater in older psoriatic subjects than in controls.
Mild and moderate psoriatic disease occurred in 79% (n=255,285) and 21% (n=66,069) of patients with psoriatic disease, respectively, including the incidence of UC, CD, RA, SLE, alopecia areata, and type diabetes. 1, and AS were greater compared to controls.
After controlling for age, gender, CCI, and insurance type, patients with moderate to severe psoriatic disease showed a significantly increased risk of rheumatoid arthritis, AS, and type 1 diabetes, with aHR values of 1.5, 1.5, and 1.2, respectively.
Conclusion
Based on the study results, patients with psoriatic conditions have a greater chance of developing autoimmune diseases.
This study also found the occurrence of autoimmune diseases unrelated to psoriasis, such as Graves’ disease, Hashimoto’s disease, Sjögren’s syndrome, and systemic sclerosis. Due to the low absolute risk, routine screening for these conditions may not be recommended.
However, appropriate investigations may be necessary in individuals with psoriatic disease to determine the existence of concomitant alopecia areata and provide tailored therapy.