All participants (demographic and anthropometric data are shown in Table 1) successfully completed the experimental sessions without side effects, except for some cases of mild tingling in the fingers at the end of the measurements. Regarding RTD, three men had to be excluded from the data analyzes because the occlusion of arterial blood flow was not possible with the MS and/or LS cuff due to the painful pinching of the skin fold and the stretching of the cuff up to the yield point. In addition, one female was excluded from data analyzes for vsysRPP, SMO2and tHb because arterial blood flow was already occluded at 20% overlap using the HS cuff.
Overlap to occlusion
There was a main effect of cuff (F2.62 = 175.679, P< 0.001, theP2= 0.850) and post hoc analysis indicated that RTD was lower in the HS compared to the MS (MD = -13.06% (-16.18 to -9.93%), P< 0.001, D= 2.06) and LS cuff (MD = -23.78% (-26.90 to -20.65%), P< 0.001, D= 3.75). In addition, RTD was also lower using the MS compared to the LS cuff (MD = -10.72% (-13.85 to -7.60%), P< 0.001, D= 1.69). Descriptive data are shown in Table 2; Fig. 3.
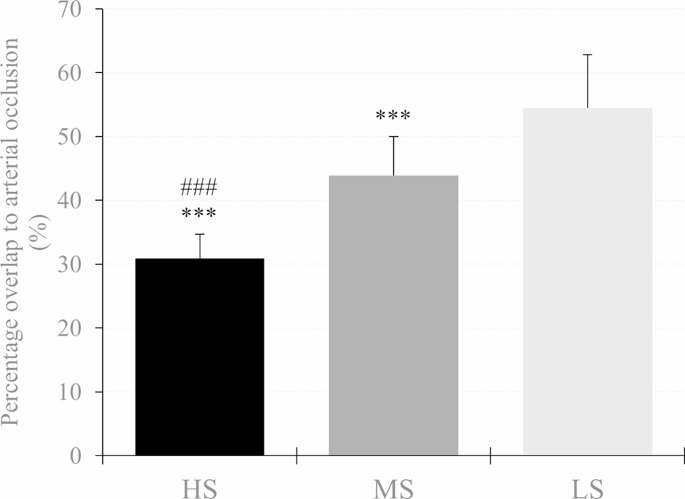
Percent overlap needed for arterial occlusion in the high stiffness (HS), medium stiffness (MS), and low stiffness (LS) cuff. A significant difference between LS and MS is shown as *p<0.05, **p < 0.01, ***p < 0.001 and #p<0.05, ##p < 0.01, ### p < 0.001 respectively
Peak systolic velocity of blood flow
There was an overlap × cuff interaction (F3,642,120,192 = 71.952, P< 0.001, theP2= 0.686) and a main effect of overlap (F1,770,58,422 = 161.427, P< 0.001, theP2= 0.830) and cuff (F2.66 = 50.380, P< 0.001, theP2= 0.604) for vsys. Post hoc analysis showed that vsys was lower at 30% overlap in each cuff compared to baseline (HS: MD = -62.67 cm s− 1(-70.97 to -54.37 cm s− 1 ), P< 0.001, D= 3.95; MS: MD = -21.53 cm s− 1(-29.83 to -13.24 cm s− 1 ), P< 0.001, D= 1.36; LS: MD = -11.02 cm s− 1(-19.31 to -2.72 cm s− 1 ), P< 0.001, D= 0.69). Furthermore, when the HS cuff was applied, vsys was also lower at 20% overlap compared to baseline (MD = -20.51 cm s− 1(-28.80 to -12.21 cm s− 1 ), P< 0.001, D= 1.29). As for cuff differences, vsys was lower at 20% and 30% overlap using the HS cuff compared to the MS (MD = -16.89 cm s− 1(-26.08 to -7.70 cm s− 1 ), P< 0.001, D= 1.07 and MD = -39.91 cm s− 1(-49.11 to -30.71 cm s− 1 ), P< 0.001, D= 2.52, respectively) and the LS cuff (MD = -17.60 cm s− 1(-26.80 to -8.41 cm s− 1 ), P< 0.001, D= 1.11 and MD = -53.99 cm s− 1(-63.18 to -44.79 cm s− 1 ), P< 0.001, D= 3.40 respectively). Furthermore vsys was also lower at 30% overlap using the MS compared to the LS cuff (MD = -14.08 cm s− 1(-23.27 to -4.88 cm s− 1), P< 0.001, D= 0.89). Descriptive data are shown in Table 2; Fig. 4.
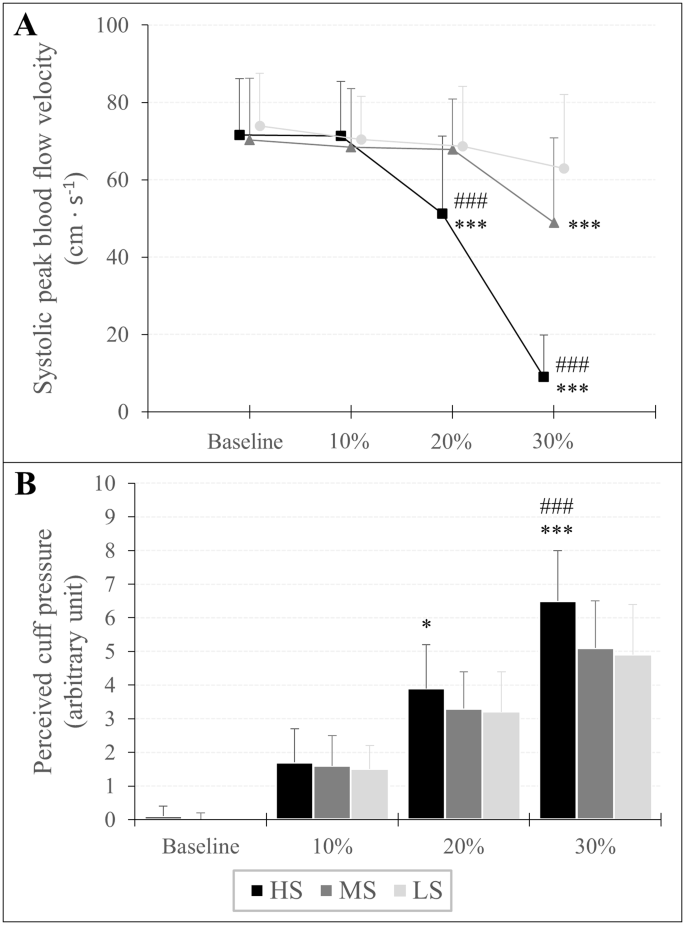
Peak systolic velocity of blood flow (A) and assessment of perceived cuff pressure (B) in response to progressive practical pressure on blood flow restriction, expressed as percent overlap in relation to the individual’s arm circumference. A significant difference between LS and MS is shown as *p<0.05, **p < 0.01, ***p < 0.001 and #p<0.05, ##p < 0.01, ###p < 0.001 respectively
Assessment of perceived cuff pressure
An overlap × cuff interaction (F3,946,130,209 = 13.994, P< 0.001, theP2= 0.298) and a main effect of overlap (F1,668,55,046 = 674.771, P< 0.001, theP2= 0.953) and cuff (F2.66 = 11.067, P< 0.001, theP2= 0.251) was found for RPP. A post hoc analysis showed that RPP increased at each %overlap stage compared to baseline for all three cuffs (HS10%: MD = 1.57 au (0.98 to 2.17 au), P< 0.001, D= 1.51; HS20%: MD = 3.80 au (3.21 to 4.40 au), P< 0.001, D= 3.66; HS30%: MD = 6.41 au (5.82 to 7.00 au), P< 0.001, D= 6.16; MS10%: MD = 1.54 au (0.94 to 2.13 au), P< 0.001, D= 1.48; MS20%: MD = 3.32 au (2.72 to 3.91 au), P< 0.001, D= 3.19; MS30%: MD = 5.03 au (4.44 to 5.63 au), P< 0.001, D= 4.84; LS10%: MD = 1.46 au (0.87 to 2.05 au), P< 0.001, D= 1.40; LS20%: MD = 3.18 au (2.59 to 3.77 au), P< 0.001, D= 3.05; LS30%: MD = 4.95 au (4.36 to 5.55 au), P< 0.001, D= 4.76). Regarding differences between cuffs, RPP was higher using the HS cuff with 20% overlap compared to the LS cuff (MD = 0.71 au (0.06 to 1.37 au), P= 0.016, D= 0.69) and with an overlap of 30% compared to Member States (MD = 1.43 (0.78 to 2.09 au), P< 0.001, D= 1.38) and LS cuff (MD = 1.54 au (0.89 to 2.19 au), P< 0.001, D= 1.48). Descriptive data are shown in Table 2; Fig. 4.
Oxygenation of the muscles
SMO2: There was an overlap × cuff interaction (F2,374,78,326 = 3.232, P= 0.037, theP2= 0.089) and a main effect of overlap (F1,297,42,808 = 404,914.= P< 0.001, theP2= 0.925) and gender (F1.33 = 5.096, P= 0.031, theP2= 0.134) for SMO2. Post hoc analysis showed that SMO2 was lower at 20% overlap (HS: MD = -9.94% (-12.43 to -7.45%), P< 0.001, D= 1.15; MS = -8.28% (-10.77 to -5.78%), P< 0.001, D= 0.96; LS: MD = -7.42% (-9.98 to -5.00%), P< 0.001, D= 0.87) and 30% overlap (HS: MD = -17.46% (-19.95 to -14.97%), P< 0.001, D= 2.02; MS: MD = -14.91% (-17.42 to -12.42%), P< 0.001, D= 1.72; LS: MD = -13.79% (-16.28 to -11.30%), P< 0.001, D= 1.59) compared to baseline. Furthermore, the main effect of sex indicated that regardless of overlap and cuff used, SMO2 was lower in men than in women (MD = -5.16% (-9.80 to -0.51%), P= 0.031, D= 0.60).
tHb: An overlap × cuff (F3,072,101,386 = 6,440, P< 0.001, theP2= 0.163) and overlap × sex interaction (F1,187,39,158 = 14.814, P< 0.001, theP2= 0.310) and a main effect of overlap (F1,187,39,158 = 117,125.= P< 0.001, theP2= 0.780) and gender (F1.33 = 27.981, P< 0.001, theP2= 0.459) was found for tHb. Post hoc tests showed that tHb was higher at 20% overlap (HS: MD = 0.13 au (0.09 to 0.18 au), P< 0.001, D= 0.45; MS: MD = 0.10 au (0.05 to 0.14 au), P< 0.001, D= 0.32; LS: MD = 0.06 au (0.02 to 0.11 au), P< 0.001, D= 0.20) and 30% overlap (HS: MD = 0.22 au (0.18 to 0.27 au), P< 0.001, D= 0.75; MS: MD = 0.17 au (0.12 to 0.21 au), P< 0.001, D= 0.55; LS: MD = 0.15 au (0.10 to 0.19 au), P< 0.001, D= 0.45) compared to baseline. Moreover, tHb was already higher at a 10% overlap using the HS (MD = 0.05 au (0.00 to 0.09 au), P= 0.018, D= 0.16) compared to baseline. Regarding sex differences, post hoc analysis showed that regardless of cuff, tHb was higher by 10% (MD = 0.06 au (0.01 to 0.10 au), P= 0.003, D= 0.20), 20% (MD = 0.15 au (0.11 to 0.20 au), P< 0.001, D= 0.52) and 30% overlap (MD = 0.24 au (0.19 to 0.29 au), P< 0.001, D= 0.80) in men, while in women tHb was only higher during a 30% overlap compared to baseline (MD = 0.12 au (0.07 to 0.16 au), P< 0.001, D= 0.39). In addition, tHb was higher in men compared to women at baseline (MD = 0.51 au (0.31 to 0.70 au), P< 0.001, D= 1.69). Descriptive data are shown in Table 2.


 Bunions can be a real pain to deal with on your own. The symptoms of a bunion can be very disabling, making it difficult to walk or get from one place to another. The good news is that some treatment options can help you repair your bunion. With the right approach, your bunion problem can be a thing of the past. A lapiplasty is an approach that many people use to treat their bunion problems. Let’s take a closer look at what exactly a lapiplasty is and how it can help you say goodbye to bunion
Bunions can be a real pain to deal with on your own. The symptoms of a bunion can be very disabling, making it difficult to walk or get from one place to another. The good news is that some treatment options can help you repair your bunion. With the right approach, your bunion problem can be a thing of the past. A lapiplasty is an approach that many people use to treat their bunion problems. Let’s take a closer look at what exactly a lapiplasty is and how it can help you say goodbye to bunion